|
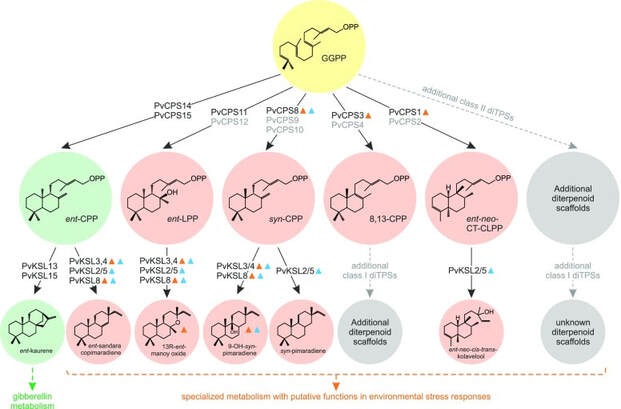
Of the myriad specialized metabolites that plants deploy to adapt to environmental challenges, terpenes form the largest group. In many major crops, unique terpene blends serve as key stress defenses that directly impact plant fitness and yield. In addition, terpenes, such as bisabolene and pinene, are used for biofuel manufacture. Thus, engineering of terpene metabolism provides a versatile resource for advancing biofuel feedstock production, but requires a system-wide knowledge of the diverse biosynthetic machinery and defensive potential of often species-specific terpene blends. This project would merge genome-wide enzyme discovery with comparative –omics, protein structural and plant microbiome studies to define the biosynthesis and stress-defensive functions of switchgrass (Panicum virgatum) terpene metabolism. These insights would be combined with the development of non-transgenic genome editing tools to design plants with desirable terpene blends for improved biofuel production on marginal lands. As a dedicated lignocellulosic biofuel feedstock with high net energy yield, stress tolerance, and available genome resources, switchgrass is well-suited for devising new avenues for U.S. biofuel production. Presently, we have identified several stress-related diterpene synthases (diTPSs) in switchgrass. Mining of the allotetraploid switchgrass genome identified an expansive diTPS family of 31 members, and biochemical analysis of 11 diTPSs revealed a modular metabolic network producing a diverse array of diterpenoid metabolites. In addition to ent-copalyl diphosphate (CPP) and ent-kaurene synthases predictably involved in gibberellin biosynthesis, we identified syn-CPP and ent-labda-13-en-8-ol diphosphate (LPP) synthases as well as two diTPSs forming (+)-labda-8,13E-dienyl diphosphate (8,13-CPP) and ent-neo-cis-trans-clerodienyl diphosphate (CT-CLPP) scaffolds not observed previously in plants. Structure-guided mutagenesis of the (+)-8,13-CPP and ent-neo-CT-CLPP synthases revealed residue substitutions in the active sites that altered product outcome, representing potential neofunctionalization events that occurred during diversification of the switchgrass diTPS family. The conversion of ent-CPP, ent-LPP, syn-CPP, and ent-neo-CT-CLPP by promiscuous diTPSs further yielded distinct labdane-type diterpene olefins and alcohols. Of these metabolites, the formation of 9β-hydroxy-syn-pimar-15-ene and the expression of the corresponding genes were induced in roots and leaves in response to oxidative stress and ultraviolet irradiation, indicating their possible roles in abiotic stress adaptation. Together, these findings expand the known chemical space of diterpenoid metabolism in monocot crops toward systematically investigating and ultimately improving stress resilience traits in crop species.
|
Select project publications |