|
Expansive gene duplication and sub/neo-functionalization events are key factors driving the rapid divergence of terpenoid metabolism. These evolutionary events have been aided by the extensive functional plasticity of terpene synthases (TPS) and cytochrome P450 monooxygenases (P450), core enzymes in generating terpenoid chemical diversity, where minor active site alterations can dramatically impact product outcome, thus enabling the emergence of new functions with minimal investment in evolving new enzymes. Hence, there has been a long-standing interest in deciphering the mechanisms underlying TPS and P450 catalysis and substrate/product specificity. Combining protein crystallization and homology modeling with structure-guided site-directed mutagenesis approaches, we work to provide mechanistic insight into the catalytic specificity of TPS and P450 enzymes and apply this knowledge to protein engineering for advancing the synthetic biology platforms for terpenoid bioproduct manufacture.
|
Example: Modulation of stereospecificity in clerodane-metabolic TPS from switchgrass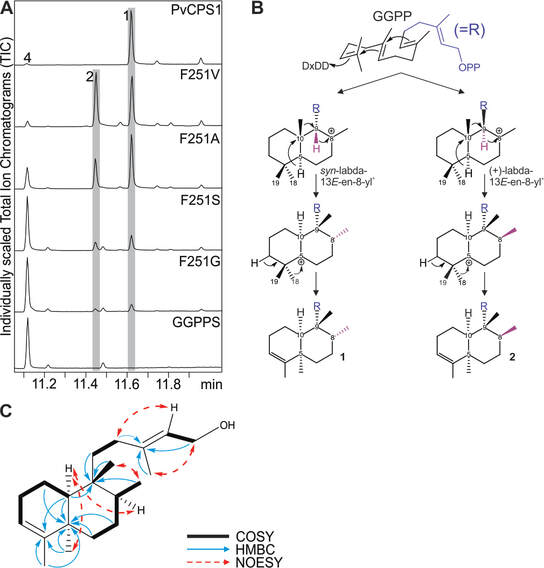
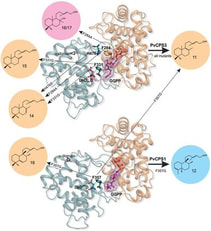
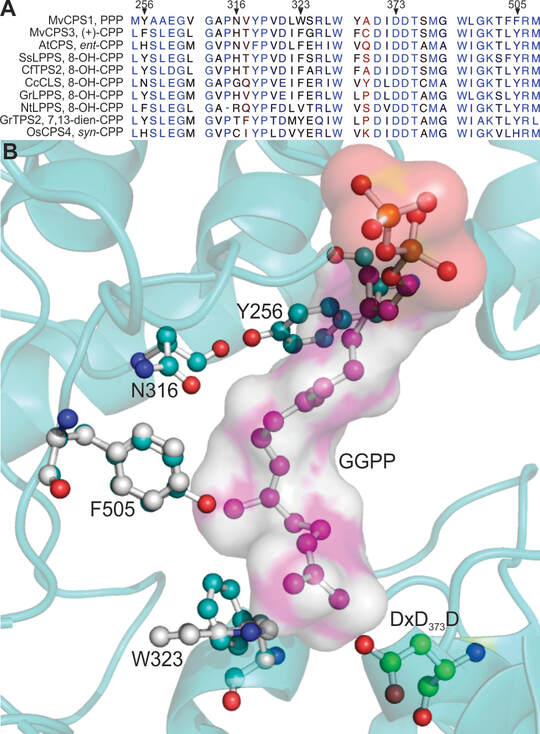
The diterpene synthase clerodienyl diphosphate synthase 1 (PvCPS1) from the crop plant switchgrass (Panicum virgatum) stereoselectively converts (E,E,E)-geranylgeranyl diphosphate (GGPP) into the clerodane natural product, cis-trans-clerodienyl diphosphate (CLPP, 1). Structure-guided point mutations of PvCPS1 redirected product stereoselectivity toward the formation of a rare cis-clerodane diastereomer, cis-cis-CLPP (2). These results support the hypothesis that substrate configuration and minor active-site alterations impact precatalysis substrate folding in the stereoselective biosynthesis of clerodane diterpenoid scaffolds, and can be employed to provide enzymatic access to a broader range of bioactive clerodane natural products.
Publication: Pelot et al. (2019) Chembiochem 20:111-17 |
|
Select other project-related publications |