Modular biochemical networks of maize anti-pathogen defense
Researched through integrating synthetic biochemistry, genetic and physiological function
In collaboration with Dr. Eric Schemlz at UC San Diego |
|
Extremely diverse and largely unresolved plant specialized metabolites defend crop plants against pests, pathogens and other stresses. Additionally, complex combinations of biotic and abiotic stresses hamper defense capacities, causing expanded pathogen susceptibility and yield losses. Essential to the understanding and agricultural optimization of plant resilience is a fundamental knowledge of the metabolic networks that govern innate immunity and vigor.
As the world's largest annually harvested crop, maize (Zea mays) is protected by an array of functionalized diterpenoids that are formed specifically to impede pathogen progression at sites of attack. Recent research advances highlight the opportunity and value of precisely examining the genetic, biochemical and functional breadth of maize biochemical defenses. This collaborative project leverages existing complementary approaches to unravel fungal-elicited maize terpenoid biosynthesis and pathogen resistance relationships using both in vitro and in vivo pathway discovery to address prevalent knowledge gaps. This project will provide critical knowledge on biochemical mechanisms mediating maize disease resistance through 3 specific aims:
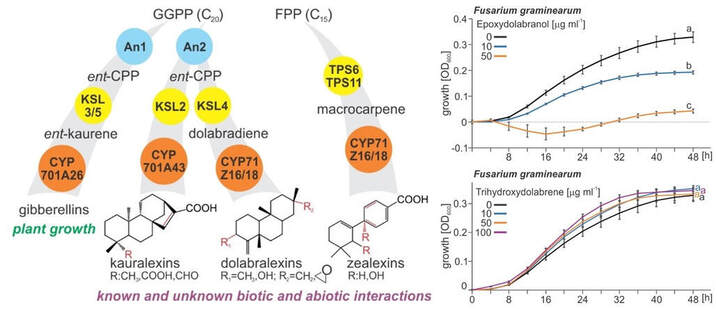
For example, we have identified a new class of maize diterpenoids, named dolabralexins (see above). Dolabralexin biosynthesis involves the sequential activity of two diterpene synthases, the ent-copalyl diphosphate synthase (ZmAN2) and kaurene synthase-like 4 (ZmKSL4). In addition, we biochemically characterized a cytochrome P450 monooxygenase, ZmCYP71Z16, which catalyzes the oxygenation of dolabradiene to afford the epoxides 15,16-epoxy-dolabradiene and 3β-epoxydolabranol. Absence of dolabradiene and 3β-epoxydolabranol in ZmAN2 mutants under elicited conditions confirms the in vivo biosynthetic requirement of ZmAN2. Combined mass spectrometry and nuclear magnetic resonance experiments demonstrate that much of the 3β-epoxy-dolabranol is further converted into 3β,15,16-trihydroxydolabrene (THD).
Metabolite profiling of common maize cultivars under field conditions supports the widespread biosynthesis of dolabralexins in roots with THD as the predominant diterpenoid accumulating. Oxidative stress induces dolabralexin accumulation and transcript expression of ZmAN2 and ZmKSL4 in root tissues. Likewise, metabolite and transcript accumulation are up-regulated in response to elicitation with the fungal pathogens Fusarium verticillioides and F. graminearum. Consistently, 3β-epoxydolabranol and THD significantly inhibit growth of both pathogens in vitro at 10 µg/mL, although THD-mediated inhibition is specific to F. verticillioides. These findings support defense-related roles for dolabralexins in maize stress interactions and expand the known chemical space of diterpenoid defenses as genetic targets for understanding and ultimately improving maize resilience. |
select project publications |